
Exogenic Geomorphic Movements: Denudation & Weathering
Subscribe to Never Miss an Important Update! Assured Discounts on New Products!
Must Join PMF IAS Telegram Channel & PMF IAS History Telegram Channel
Last updated on April 26, 2024 11:37 PM
Exogenic Geomorphic Movements
- The geomorphic processes on the earth’s crust or its surface brought down by the forces emanating from above the earth’s surface (wind, water) are called exogenic geomorphic process.
- Exogenic geomorphic process gives rise to exogenic geomorphic movements or simply exogenic movements such as weathering and erosion.
- The effects of most of the exogenic geomorphic processes are small and slow but will, in the long run, affect the rocks severely due to continued fatigue.
The force behind Exogenic Movements
- Exogenic processes are a direct result of the sun’s heat.
- Sun’s energy dictates the weather patterns like winds, precipitation, etc.
- Sun’s heat along with weather patterns are responsible for stress-induced in earth materials giving rise to exogenic movements (weathering and erosion).
- Earth materials become subjected to molecular stresses caused due to temperature changes.
- Chemical processes normally lead to loosening of bonds between grains.
- Stress is produced in a solid by pushing or pulling (shear stresses ― separating forces) forces.
Watch the video for quick and better understanding
Denudation
- All the exogenic processes (weathering and erosion) are covered under a general term, denudation.
- The word ‘denude’ means to strip off or to uncover.
- Denudation depends on physical (folds, faults, orientation and inclination of beds, presence or absence of joints, bedding planes, hardness or softness of constituent minerals, permeability) and chemical (chemical susceptibility of mineral constituents to corrosion) properties of the rocks.
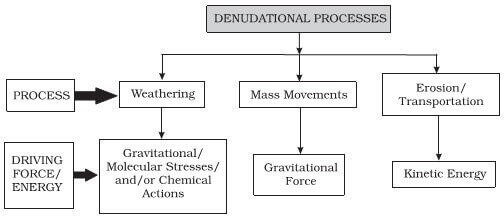
Weathering
- Weathering is the disintegration of rocks, soil, and minerals under the influence of physical (heat, pressure) and chemical (leaching, oxidation and reduction, hydration) agents.
- As very little or no motion of materials takes place in weathering, it is an in-situ or on-site process.
- The weathered material is carried farther away by erosion.
- There are three major groups of weathering processes: 1) chemical; 2) physical or mechanical; 3) biological weathering processes. All the types of weathering often go hand in hand.
Physical Weathering Processes
- Physical weathering involves mechanical disintegration of rocks due to temperature changes, freeze-thaw cycles, wet-dry cycles, crystallisation of salts, animal and plant activity, etc.
- Various mechanisms of physical weathering are explained below.
Exfoliation due to pressure release or unloading
- Intrusive igneous rocks formed deep beneath the Earth’s surface are under tremendous pressure due to overlying load.
- Removal of the overlying load because of continued erosion causes vertical pressure release with the result that the upper layers of the rock expand and fracture parallel to the surface.
- Over time, sheets of rock break away from the exposed rocks along the fractures, a process known as exfoliation.
- Exfoliation due to pressure release is also known as “sheeting“.

Exfoliation due to thermal stress weathering
- Thermal stress weathering results from the subsequent expansion and contraction of rocks caused by diurnal and seasonal variations in the temperatures.
- The surface layers of the rocks tend to expand more than the rock at depth, and this leads to peeling off of the surface layers (exfoliation).
- This process is most effective in dry climates and high elevations where diurnal temperature changes are drastic.
- Although temperature changes are the principal driver, moisture can enhance thermal expansion in rock.
Granular Disintegration
- Granular disintegration happens in rocks composed of different types of coarse-grained minerals.
- Dark-coloured minerals absorb more heat than the light-coloured minerals.
- This leads to differential expansion and contraction of mineral grains resulting in grain by grain separation from the rock.

Frost weathering
- During the warm season, the water penetrates the pore spaces or fractures in rocks.
- During the cold season, the water freezes into ice, and its volume expands as a result.
- This exerts tremendous pressure on rock walls to tear apart even where the rocks are massive.
- Frost weathering occurs due to the growth of ice within pores and cracks of rocks during repeated cycles of freezing and melting.
- Frost weathering is the collective name for several processes where ice is present.
- These processes include frost shattering, frost-wedging and freeze-thaw weathering.
Frost wedging
- Freeze wedging is caused by the repeated freeze-thaw cycle.
- Cracks filled with water are forced further apart with subsequent freezing and thawing.

Shattering
- Severe frost can disintegrate rocks along weak zones to produce highly angular pieces with sharp corners and edges through the process of shattering.
- Shattering piles up rock fragments called scree at the foot of mountain areas or along slopes.

Block Separation (freeze-thaw weathering)
- Repeated freeze-thaw cycles weaken the rocks which, over time, break up along the joints into angular pieces. The splitting of rocks along the joints into blocks is called block disintegration.

Salt Weathering
- Salt weathering occurs when saline solutions seep into cracks and joints in the rocks and evaporate, leaving salt crystals behind.
- Salt crystals expand during the crystallization process and also when they are subjected to above normal temperatures.
- The expansion in near-surface pores causes splitting of individual grains within rocks, which eventually fall off (granular disintegration or granular foliation).
- Salt weathering is normally associated with arid climates where strong heating causes strong evaporation and crystallisation.
Mass Wasting
- Mass wasting is the mass movement of unconsolidated soil, sand, rocks, regolith (the layer of unconsolidated solid material covering the bedrock of a planet), etc. along a slope under the influence of gravity.
- Mass wasting occurs when the gravitational force acting on a slope exceeds its resisting force leading to slope failure (mass wasting).
- Timescales of the mass wasting process may be a few seconds (debris flows and mudflows) or hundreds of years (mass wasting along the slopes of stable mountains leaving behind alluvial fan like structures).

Chemical Weathering
- Chemical weathering involves chemical decomposition of rocks and soil.
- Chemical weathering processes include dissolution, solution, carbonation, hydration, oxidation and reduction that act on the rocks to decompose, dissolve or reduce them to a fine state.
- These weathering processes are interrelated and go hand in hand and hasten the weathering process.
- Acids produced by microbial and plant-root metabolism, water and air (oxygen and carbon dioxide) along with heat speed up all chemical reactions.
Natural dissolution
- Dissolution: a process where a solute in gaseous, liquid, or solid phase dissolves in a solvent to form a solution.
- Some minerals, due to their natural solubility (like nitrates, sulphates, and potassium), oxidation potential (iron-rich minerals) will weather through dissolution naturally (rains).
- These minerals are easily leached out without leaving any residue and accumulate in dry regions.
Solution weathering
- Solution weathering occurs when the solvent is an acidic solution rather than simple water.
- A solution is a liquid mixture in which the minor component (the solute) is uniformly distributed within the major component (the solvent).
- Acidic solutions are any solution that has a higher concentration of hydrogen ions than water; solutions that have a lower concentration of hydrogen ions than water is called basic or alkaline solutions.
Carbonation – Natural solution weathering
- Carbonation refers to reactions of carbon dioxide to give carbonates, bicarbonates, and carbonic acid.
- Carbonation weathering is a process in which atmospheric carbon dioxide leads to solution weathering.
- As rain falls, it dissolves small amounts of carbon dioxide from the air, forming a weak acid that can dissolve some minerals like limestone (calcium carbonate) (solution weathering).
- When carbonic acid reacts with limestone, it produces calcium bicarbonate, which is partially soluble in water (dissolution weathering).
- Caves are formed when underground water containing carbonic acid travels through blocks of limestone, dissolves out the limestone, and leaves empty pockets (caves) behind (E.g. Karst topography).
- Carbonation process speeds up with a decrease in temperature because colder water holds more dissolved carbon dioxide gas. Carbonation is, therefore, a large feature of glacial weathering.
Anthropogenic solution weathering
- Rainfall is naturally acidic ― pH of ~5.6 (CO2 dissolves in the rainwater producing weak carbonic acid).
- Acid rain occurs when gases such as sulphur dioxide and nitrogen oxides are present in the atmosphere.
- These oxides react in the rainwater to produce stronger acids and can lower the pH to less than 4.
- These acids are capable of attacking certain kinds of rocks in much the way that carbonic acid does.
Sulphur dioxide, SO2, comes from volcanic eruptions or fossil fuels.
The conversion of metallic ores to the pure metals often results in the formation of sulphur dioxide.
Hydration
- Hydration is the chemical addition of water that involves the rigid attachment of H+ and OH- ions to the atoms and molecules of a mineral.
- When rock minerals take up water, the increased volume creates physical stresses within the rock. For example, iron oxides are converted to iron hydroxides which are larger in volume.
- Hydration is reversible, and continued repetition of this process causes fatigue in the rocks and may lead to their disintegration.
- The volume changes in minerals due to hydration will also help in physical weathering through exfoliation and granular disintegration.
Hydrolysis
- In biological hydrolysis, a water molecule is consumed to affect the separation of a larger molecule into component parts.
- In biological hydrolysis pure water reacts with silicate or carbonate minerals resulting in the complete dissolution of the original mineral (dissolution weathering).
- Biological hydrolysis is an important reaction in controlling the amount of CO2 in the atmosphere and can affect climate.
Oxidation and Reduction
- In weathering, oxidation means a combination of a mineral with oxygen to form oxides (rusting in case of iron) or hydroxides. Red soils appear red due to the presence of iron oxides.
- Oxidation occurs where there is ready access to the atmosphere and water.
- The minerals most commonly involved in this process are iron, manganese, sulphur etc.
- When oxidised minerals are placed in an environment where oxygen is absent, reduction takes place.
- Such conditions usually exist below the water table, in areas of stagnant water and waterlogged ground.
- The red colour of iron upon reduction turns to greenish or bluish grey.
Biological activity and weathering
- Biological weathering is the removal of minerals from the environment due to growth or movement of organisms.
- Living organisms contribute to both mechanical and chemical weathering.
- Lichens and mosses grow on essentially bare rock surfaces and create a more humid chemical microenvironment.
- On a larger scale, seedlings sprouting in a crevice and plant roots exert physical pressure as well as providing a pathway for water and chemical infiltration.
- Burrowing and wedging by organisms like earthworms, rodents etc., help in exposing the new surfaces to chemical attack and assists in the penetration of moisture and air.
- Decaying plant and animal matter help in the production of humic, carbonic and other acids which enhance decay and solubility of some elements.
- Algae utilise mineral nutrients for growth and help in the concentration of iron and manganese oxides.
Significance of weathering
- Weathering is the first step in the formation of soil from rocks.
- Weathering weakens soil and rocks and makes it easy to exploit natural resources.
- Weathering leads to natural soil enrichment.
- Weathering leads to mineral enrichment of certain ores by leaching unwanted minerals leaving behind the valuable ones.
Soil Erosion, another exogenic movement, is covered in the subsequent chapters.
Last updated on April 26, 2024 11:37 PM




![PMF IAS Environment for UPSC 2022-23 [paperback] PMF IAS [Nov 30, 2021]…](https://pmfias.b-cdn.net/wp-content/uploads/2024/04/pmfiasenvironmentforupsc2022-23paperbackpmfiasnov302021.jpg)






Good Evening Sir,
I am not getting the article explaining erosion and its type,can you please direct me to the link.
Thank you sir.
http://www.pmfias.com/soil-degradation-soil-erosion-deforestation-overgrazing-faulty-methods-of-agriculture-salinity-alkalinity-desertification-waterlogging/
Must Detailed Explaintion of Erosion Will be given under Environment Notes [Will be upload on Instamojo by May 5th]
Okay,Thankyou sir.
Will you be provoding notes for Agriculture in Indian geography as it is an important part for prelims.
Please notify if possible.
Yes. By the End of May
Okay,please make sure you provide them in insta or site..as I might be dependent on them.
Thanx.
Landslides are covered in geomorphology
I am not able to get landslides notes. Please provide the direct link