
Adiabatic Lapse Rate | Latent Heat of Condensation
Subscribe to Never Miss an Important Update! Assured Discounts on New Products!
Must Join PMF IAS Telegram Channel & PMF IAS History Telegram Channel
Condensation of water vapour, thunderstorms, cyclonic and anticyclonic conditions etc. depend on Adiabatic Lapse Rate. Adiabatic Lapse Rate determines the Rate Of Condensation and the rate of condensation determines the amount of Latent Heat of Condensation Released.
All the explanation below is meant to explain ‘Adiabatic Lapse Rate’ and ‘Latent Heat of Condensation’. These two terms occur frequently in almost all the future topics of climatology. They won’t be specifically asked in the exam. But understanding them once for all will help immensely in understanding the future posts on climatology.
Lapse Rate
- Lapse rate is rate of change in temperature observed while moving upward through the Earth’s atmosphere (troposphere to be specific).
- The lapse rate is considered positive when the temperature decreases with elevation, zero when the temperature is constant with elevation, and negative when the temperature increases with elevation (temperature inversion).
- The lapse rate of non-rising air – commonly referred to as the normal, or Environmental, Lapse Rate (ELR) – is highly variable, being affected by radiation, convection, and condensation; it averages about 5 °C per kilometer in the lower atmosphere (troposphere).
Why does temperature fall with elevation
- When we move up a hill, we notice a fall in temperature. This fall in temperature with elevation is primarily due to two reasons.
- With increase in elevation, the atmospheric pressure falls. Fall in pressure implies that the temperature also falls [Pressure is directly proportional to Temperature and vice versa]
- With increase in elevation, the concentration of greenhouse gases decrease (Water vapor and carbon dioxide fall sharply with elevation). Hence the heat absorption capacity of atmosphere will also decrease.
This sort of fall in temperature with elevation is called Temperature Lapse and the rate at which it happens is called Temperature Lapse Rate or simply Lapse rate.
Adiabatic Lapse rate
- Lapse rate is the rate of fall in temperature of atmosphere with elevation.
- Adiabatic Lapse Rate is the rate of fall in temperature of a rising or a falling air parcel adiabatically.
- Adiabatic or adiabatically: Heat doesn’t enter or leave the system. All temperature changes are internal.
- Adiabatic Lapse rate is governed by Gas law.
Gas law
- According to gas law Pressure ‘P’ is directly proportional to Temperature ‘T’ when Volume ‘V’ is a constant.
Just for understanding
Example 1: A balloon
- When we blow air into a balloon, pressure increases but temperature doesn’t increase due to proportionate increase in volume (here V is not constant). When excess air is blown, balloon bursts as it cannot with stand the pressure.
Example 2: Vehicle tube
- In a vehicle tube, volume remains constant. When air is blown, pressure increases and hence the temperature.
- We are usually advised not to have full blown tubes because when vehicle travels on a road, the friction between the tire and the road increases the temperature of the air in the tube. As temperature is directly proportional to pressure, increase in temperature leads to increase in pressure and at certain pressure threshold, the tire bursts.
The above examples explain the relation between Pressure, Temperature and Volume. They are both non-adiabatic processes as there is (will be) heat exchange between the system and the external environment.
Adiabatic Process: A Parcel of Rising or Falling Air
- An air bubble rises in water whereas stone sinks. This is obvious. The stone is denser (heavier than water) and it sinks whereas the air bubble is less denser (lighter than water) and it rises.
- Similarly, a parcel of air rises when it is less denser than the surrounding environment and it falls when its density becomes greater than the surrounding environment.
A Parcel of Rising Air
- When an air parcel is subjected to differential heating compared to the surrounding air, it becomes lighter (less denser) or heavier (more denser) depending on whether the air parcel is heated or cooled.
- When an air parcel receives more heat than the surrounding air, its temperature increases leading to an increase in volume (Increase in Volume == Fall in Density). The air parcel becomes lighter than the surrounding air and it starts to rise. This process is non-adiabatic (there is heat exchange between the air parcel and the external environment).
- But when the air parcel starts to rise, the ambient pressure on it starts to fall [The atmospheric pressure decreases with height, so the pressure on the air parcel decreases with height]. With the fall in ambient pressure, the temperature falls and the volume increases. This is adiabatic [there is no heat exchange between the air parcel and the external environment. All the temperature changes are internal. Temperature changes are only due to change in pressure or volume or both].
- This fall in temperature with the rising of the air parcel is called Adiabatic Temperature Lapse. And the rate at which it happens is called Adiabatic Lapse Rate [This is Positive Adiabatic Lapse Rate as the Temperature is falling].
[Lapse Rate == fall in temperature with height. Adiabatic Lapse Rate == Fall in temperature in a rising parcel of air without losing any internal heat]
Rising of a parcel of air (and associated Positive Adiabatic Lapse Rate) is the first step in the formation of Thunderstorms, Tornadoes and Cyclones. [We will see this in detail in future posts]
A Parcel of Falling Air
- An air parcels falls to the lower levels of troposphere when it is cooled sufficiently.
- When an air parcel is in the upper levels, it gets cooled due to lower temperatures (Lapse Rate). It’s volume falls and it’s density increases. When it becomes more denser than the surroundings, it starts to fall.
- This also happens when an air parcel is in contact with cooler surfaces like mountain slopes. We will see more about this in temperature inversion.
- The beginning of fall is a non-adiabatic process as there is an exchange of heat between the air parcel and the surrounding environment.
- When an air parcel is falling, the atmospheric pressure acting on it will increase and its internal temperature will increase adiabatically. [This is Negative Adiabatic Lapse Rate as the Temperature is rising].
Katabatic Wind is a hot dry wind that blows down a mountain slope. It is an example for a falling parcel of air in which the temperature changes happen adiabatically.
Adiabatic Lapse Rate in simple terms
- Adiabatic change refers to the change in temperature with pressure.
- On descent through atmosphere, the lower layers are compressed under atmospheric pressure. As a result, the temperature increases.
- On ascent, the air expands as pressure ‘decreases’. This expansion reduces the temperature and aids condensation of water vapour. Condensation of water vapour releases the Latent Heat of Condensation in the process.
- This latent heat of condensation is the major driving force behind tropical cyclones, convectional rains.
Wet and Dry Adiabatic Lapse rate
- Adiabatic lapse rates are usually differentiated as dry or wet (moist).
Dry Adiabatic Lapse rate
- The Dry Adiabatic Lapse Rate (DALR) is the rate of fall in temperature with altitude for a parcel of dry or unsaturated air (air with less moisture, to keep it simple) rising under adiabatic conditions.
- Unsaturated air has less than 100% relative humidity (we will study about Humidity in future posts).
[Saturated air == The air that cannot hold any more moisture. Its stomach is full.
Unsaturated air == It’s stomach is not full. It can accommodate some more moisture.]
- When a rising air parcel has little moisture, condensation during upliftment is low, the latent heat of condensation released is low [Less additional heat from inside]. As a result, the fall in temperature with height is greater compared to Adiabatic Lapse Rate (normal parcel of air). [I have explained in detail in my videos]
- The dry adiabatic lapse rate for the Earth’s atmosphere equals 8° C per kilometre.
- Dry Adiabatic Lapse rate is mainly associated with stable conditions [because it has less moisture].
Wet Adiabatic Lapse rate
- When an air parcel that is saturated (stomach full) with water vapour rises, some of the vapour will condense and release latent heat [Additional Heat from inside]. This process causes the parcel to cool more slowly than it would if it were not saturated.
- The moist adiabatic lapse rate varies considerably because the amount of water vapour in the air is highly variable. The greater the amount of vapour, the smaller the adiabatic lapse rate [because the condensation process keeps on adding more latent heat of condensation]. On an average it is taken as 4° C per kilometre.
- Wet Adiabatic Lapse rate is mainly associated with unstable conditions [because it has more moisture].
- As an air parcel rises and cools, it may eventually lose its moisture through condensation; its lapse rate then increases and approaches the dry adiabatic value.
Significance in meteorology
- The difference between the normal lapse rate in the atmosphere and the dry and moist adiabatic lapse rates determines the vertical stability of the atmosphere.
- For this reason, the lapse rate is of prime importance to meteorologists in forecasting certain types of cloud formations, the incidence of thunderstorms, and the intensity of atmospheric turbulence.
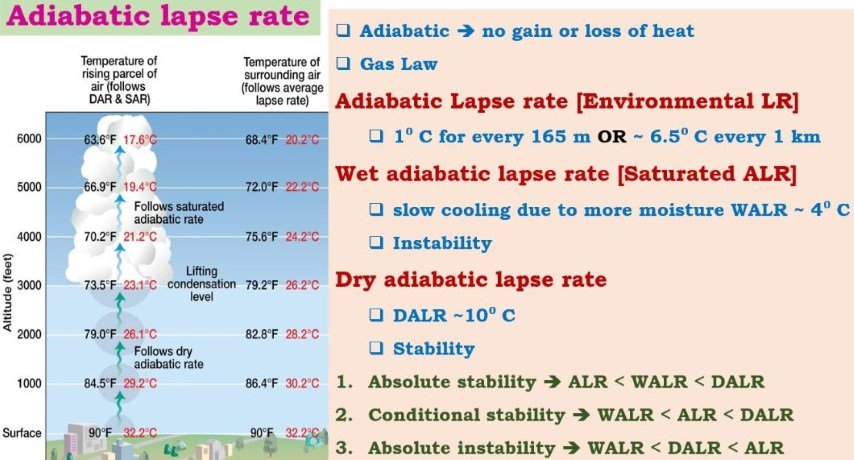
Weather conditions at different adiabatic lapse rates
- LR (Lapse Rate) = Average Adiabatic Lapse Rate of entire atmosphere = 6 °C/km [ALR of a place may be greater than or lesser than the Laspe Rate of atmosphere, i.e, it may be less than or greater than 6 °C/km]
- If ALR at a place is greater than 6 °C/km then it is called DALR = Less moisture than normal = more stable than normal.
- If ALR at a place is lesser than 6 °C/km then it is called WALR = More moisture than normal = less stable than normal or instability.
Absolute stability: ALR (at a place) > DALR == Little moisture in the air parcel == It won’t rain
Conditional stability: WALR < ALR < DALR == Normal moisture conditions == It may or may not rain
Absolute instability: ALR (at a place) < WALR == Excess moisture in the air parcel == It will rain violently.
The image needs correction.
Absolute stability: ALR (at a place) > DALR
- The above condition simply means that there is little moisture in air.
- When there is little moisture, condensation of water vapour is low, so latent of condensation released will be low, and the rising parcel of air gets cold quickly, and it falls to the ground once it becomes denser.
- So there will be no cloud formation and hence there will be no rain (thunderstorms).
- This simply means that the condition is stable.
Conditional stability: WALR < ALR < DALR
- The above condition simply means that there is enough moisture in air and there are chances of thunderstorms.
- When there is considerable moisture in the air parcel, condensation of water vapour will be reasonably high, so latent of condensation released will be adequate to drive a thunderstorm. The occurrence of thunderstorm depends on external factors. So the weather will be associated with conditional stability (it may rain or it may not rain)
Absolute instability: ALR (at a place) < WALR
- The above condition simply means that there is more moisture in air and there will be thunderstorms.
- When there is unusually high moisture in the air parcel, condensation of water vapour will be very high, so latent of condensation released will be great enough to drive a violent thunderstorm. So the weather will be associated with absolute instability.
Latent Heat of Condensation
- Latent heat of condensation is the driving force behind all tropical cyclones.
Latent Heat
- It is the heat released or absorbed during phase change.
- Latent heat, characteristic amount of energy absorbed or released by a substance during a change in its physical state that occurs without changing its temperature.
- The latent heat associated with melting a solid or freezing a liquid is called the heat of fusion; that associated with vaporizing a liquid or a solid or condensing a vapour is called the heat of vaporization.
The latent heat is normally expressed as the amount of heat (in units of joules or calories) per mole or unit mass of the substance undergoing a change of state.
- For example, when a pot of water is kept boiling, the temperature remains at 100 °C until the last drop evaporates, because all the heat being added to the liquid is absorbed as latent heat of vaporization and carried away by the escaping vapour molecules.
- Similarly, while ice melts, it remains at 0 °C, and the liquid water that is formed with the latent heat of fusion is also at 0 °C.
Explanation
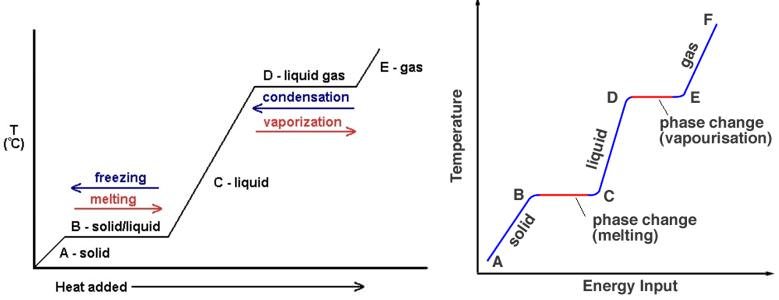
On X – axis: Heat supplied to the system.
On Y – Axis: Temperature change in the system.
- From the above graph, we can observe that there is no change in temperature in the system during change of state or phase change (solid to liquid, liquid to solid, liquid to gas and gas to liquid). Then where did the heat supplied go?
- Initially the heat supplied is used to raise the temperature of the system (A – B & C – D)
- During phase change, the heat supplied is consumed to turn water into liquid and then liquid into gas. So the heat supplied in used in phase change. Hence temperature of the system remains constant during phase change process. (B – C & D – E)
- But when gas turns into liquid or liquid into solid, heat is released. (this heat is the heat that was used during the phase change process)
So latent heat of condensation is the heat released when gases turn into liquid.
Primary References: NCERT Geography, Certificate Physical and Human Geography [Amazon and Flipkart], Spectrum’s Geography [Amazon and Flipkart] and Savindra Singh [Amazon and Flipkart]











Awesome explanation sir .. you made this look so easy.
Awesome Explanation Sir, You made it look so easy.
Thanks for your feedback bro
In the para ‘the parcel of rising air’
With the fall in ambient pressure, the temperature falls and the volume increases. This is adiabatic .
Sir,
By gas law,when pressure drops,temperature also drops but volume has to be constant.
How does a volume change occur as you have mentioned?
Gas law is just theory. But practically in many situations volume may not be constant. Let us say pressure doubled when volume is constant. Then temperature may double. Sometime volume might increase slightly due to increase in pressure. Now temperature will also increase by some extent as the increase in volume is very small.
In the paragraph ‘the rising air parcel’,
With the fall in ambient pressure, the temperature falls and the volume increases. This is adiabatic
Sir,
By gas law,volume has to be constant ,,for temperature to decrease proportionately to pressure drop.how does volume change occur?pls explain
Gas law is ideal scenario.
In practical cases, increases in temperature will lead to increase in pressure and vice versa when volume change is not proportionate.
Let us say pressure p=10 and temperature t=10 when volume v is constant.
When v is kept constant, increase in pressure will lead to increase temperature. Let us say p=20 then t=20 [increased proportionately]
Lets volume v=5, then t=15 instead of 20. There will be proportionate change [This is a raw, scientifically imperfect example]
Thank u sir.
i think in absolute stability it should be
ALR<DALR
It has been already explained .For clarity
1) WHY Adiabatic Lapse Rate must be greater than Dry Adiabatic Lapse Rate?( ALR>DALR )
Ans:In this way air won’t condense and hence won’t result in the formation of rainfall which is called INSTABILITY.
2)What is Conditional Stability?
Ans:Here WALR<ALR<DALR
in this way there is chance that DALR could be far cooler than ALR and may be it could become cooler enough to shed its weight. (It is just my imagination but lil bit confused in it )
3)What is Absolute Instability?
Ans:ALR<WALR.
The topic is somewhat confusing and it can’t be explained in few words. Just ignore it. It is not required for the exam.
i cant thank more for your site . If i qualify this exam – i know it will be bcz of this site.
Perfect explanation.