
Atmosphere: Role, Structure & Composition
Subscribe to Never Miss an Important Update! Assured Discounts on New Products!
Must Join PMF IAS Telegram Channel & PMF IAS History Telegram Channel
Last updated on April 25, 2024 1:10 PM
Atmosphere
- Our planet earth is enveloped by a deep blanket of gases extending several thousands of kilometres above its surface. This gaseous cover of the earth is known as the atmosphere.
- Like land (lithosphere) and water (hydrosphere), the atmosphere is an integral part of the earth.
- Compared to the earth’s radius, the atmosphere appears to be only a very thin layer of gases. However, because of the force of gravity, it is inseparable from the earth.
- Atmospheric pressure: The air exerts pressure on earth’s surface by virtue of its weight. This pressure is called atmospheric pressure. Atmospheric pressure is the most important climatic element. The atmospheric pressure at sea level is 1034 gm per square centimeter.
Role of Earth’s Atmosphere
- The atmosphere contains various gases like oxygen, carbon dioxide, nitrogen etc.
- Plants require carbon dioxide to survive while animals and many other organisms need oxygen for their survival. The atmosphere supplies these life giving gases.
- All life forms need a particular range of temperature and a specific range of frequencies of solar radiation to carry out their biophysical processes. The atmosphere absorbs certain frequencies and lets through some other frequencies of solar radiation. In other words, the atmosphere regulates the entry of solar radiation.
- The atmosphere also keeps the temperature over the earth’s surface within certain limits. In the absence of the atmosphere extremes of temperature would exist between day and night over the earth’s surface.
- Harmful ultraviolet radiation would find its way through, if the atmosphere (ozone in stratosphere to be specific) were absent.
- The atmosphere also takes care of extra-terrestrial objects like meteors which get burnt up while passing through the atmosphere (mesosphere to be precise) due to friction.
- Weather is another important phenomenon which dictates the direction of a number of natural and man-made processes like plant growth, agriculture, soil-formation, human settlements, etc. Various climatic factors join together to create weather.
Composition of Atmosphere
- The atmosphere is a mixture of many gases. In addition, it contains huge numbers of solid and liquid particles, collectively called ‘aerosols’.
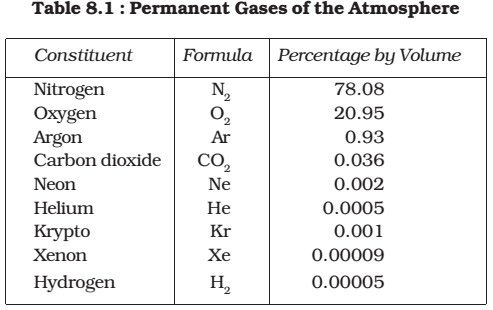
- Some of the gases may be regarded as permanent atmospheric components which remain in fixed proportion to the total gas volume.
- Other constituents vary in quantity from place to place and from time to time. If the suspended particles, water vapour and other variable gases were excluded from the atmosphere, then the dry air is very stable all over the earth up to an altitude of about 80 kilometres.
- The proportion of gases changes in the higher layers of the atmosphere in such a way that oxygen will be almost in negligible quantity at the height of 120 km. Similarly, carbon dioxide and water vapour are found only up to 90 km from the surface of the earth.
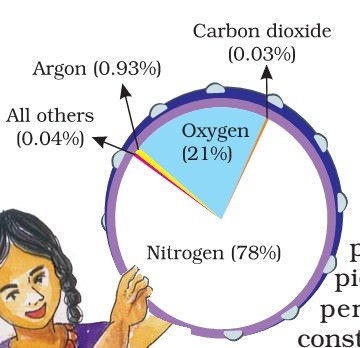
- Nitrogen and oxygen make up nearly 99% of the clean, dry air. The remaining gases are mostly inert and constitute about 1% of the atmosphere.
- Besides these gases, large quantities of water vapour and dust particles are also present in the atmosphere. These solid and liquid particles are of great climatic significance.
- Different constituents of the atmosphere, with their individual characteristics, are discussed below.
Oxygen
- Oxygen, although constituting only 21% of total volume of atmosphere, is the most important component among gases. All living organisms inhale oxygen. Besides, oxygen can combine with other elements to form important compounds, such as, oxides. Also, combustion is not possible without oxygen.
Nitrogen
- Nitrogen accounts for 78% of total atmospheric volume. It is a relatively inert gas, and is an important constituent of all organic compounds. The main function of nitrogen is to control combustion by diluting oxygen. It also indirectly helps in oxidation of different kinds.
Carbon Dioxide
- The third important gas is Carbon Dioxide which constitutes only about 03% of the dry air and is a product of combustion. Green plants, through photosynthesis, absorb carbon dioxide from the atmosphere and use it to manufacture food and keep other bio-physical processes going.
- Being an efficient absorber of heat, carbon dioxide is considered to be of great climatic significance. Carbon dioxide is considered to be a very important factor in the heat energy budget.
- With increased burning of fossil fuels – oil, coal and natural gas – the carbon dioxide percentage in the atmosphere has been increasing at an alarming rate.
- More carbon dioxide in the atmosphere means more heat absorption. This could significantly raise the temperature at lower levels of the atmosphere thus inducing drastic climatic changes.
Ozone (03)
- Ozone (03) is another important gas in the atmosphere, which is actually a type of oxygen molecule consisting of three, instead of two, atoms. It forms less than 0.00005% by volume of the atmosphere and is unevenly distributed. It is between 20 km and 25 km altitude that the greatest concentrations of ozone are found. It is formed at higher altitudes and transported downwards.
- Ozone plays a crucial role in blocking the harmful ultraviolet radiation from the sun.
- Other gases found in almost negligible quantities in the atmosphere are neon, helium, hydrogen, xenon, krypton, methane etc.
Water Vapour
- Water Vapour is one of the most variable gaseous substances present in atmosphere – constituting between 02% and 4% of the total volume (in cold dry and humid tropical climates respectively). 90% of moisture content in the atmosphere exists within 6 km of the surface of the earth. Like carbon dioxide, water vapour plays a significant role in the insulating action, of the atmosphere.
- It absorbs not only the long-wave terrestrial radiation (infrared or heat emitted by earth during nights), but also a part of the incoming solar radiation.
- Water vapour is the source of precipitation and clouds. On condensation, it releases latent heat of condensation —the ultimate driving force behind all storms.
The moisture – carrying capacity of air is directly proportional to the air temperature.
Solid Particles
- The Solid Particles present in the atmosphere consist of sand particles (from weathered rocks and also derived from volcanic ash), pollen grains, small organisms, soot, ocean salts; the upper layers of the atmosphere may even have fragments of meteors which got burnt up in the atmosphere. These solid particles perform the function of absorbing, reflecting and scattering the radiation.
- The solid particles are, consequently, responsible for the orange and red colours at sunset and sunrise and for the length of dawn (the first appearance of light in the sky before sunrise) and twilight (the soft glowing light from the sky when the sun is below the horizon, caused by the reflection of the sun’s rays by the atmosphere. Dusk: the darker stage of twilight.). The blue colour of the sky is also due to selective scattering by dust particles.
- Some of the dust particles are hygroscopic (i.e. readily absorbing moisture from air) in character, and as such, act as nuclei of condensation. Thus, dust particles are an important contributory factor in the formation of clouds, fog and hailstones.
Major Greenhouse Gases
Carbon dioxide
- Carbon dioxide is meteorologically a very important gas as it is transparent to the incoming solar radiation but opaque to the outgoing terrestrial radiation. It absorbs a part of terrestrial radiation and reflects back some part of it towards the earth’s surface. It is largely responsible for the greenhouse effect.
Ozone
- Ozone is another important greenhouse gas. But it is very small proportions at the surface.
Water vapour
- Water vapour is also a variable gas in the atmosphere, which decreases with altitude. Water vapour also decreases from the equator towards the poles.
- In the warm and wet tropics, it may account for four per cent of the air by volume, while in the dry and cold areas of desert and polar regions, it may be less than one per cent of the air.
- It also absorbs parts of the insolation from the sun and preserves the earth’s radiated heat.
- It thus, acts like a blanket allowing the earth neither to become too cold nor too hot. Water vapour also contributes to the stability and instability in the air.
Methane
- One of the most important greenhouse gases. It is produced from decomposition of animal wastes and biological matter.
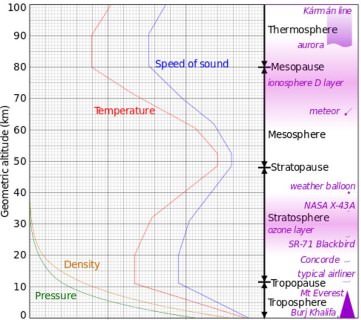
Structure of Atmosphere
- The atmosphere can be studied as a layered entity – each layer having its own peculiar characteristics. These layers are systematically discussed below.
Troposphere
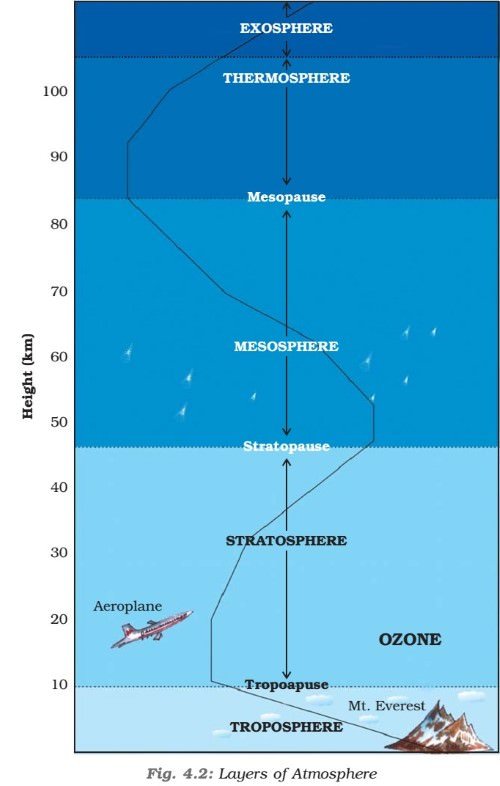
- It is the atmospheric layer between the earth’s surface and an altitude of 8 km at the poles and 18 km at the equator.
- The thickness is greater at the equator, because the heated air rises to greater heights.
- The troposphere ends with the Tropopause.
- The temperature in this layer, as one goes upwards, falls at the rate of 5°C per kilometer, and reaches -45°C at the poles and -80°C over the equator at Tropopause (greater fall in temperature above equator is because of the greater thickness of troposphere – 18 km).
- The fall in temperature is called ‘lapse rate’. (more about this in future posts)
- The troposphere is marked by temperature inversion, turbulence and eddies.
- It is also meteorologically the most significant zone in the entire atmosphere (Almost all the weather phenomena like rainfall, fog and hailstorm etc. are confined to this layer).
- It is also called the convective region, since all convection stops at Tropopause.
- The troposphere is the theatre for weather because all cyclones, anticyclones, storms and precipitation occur here, as all water vapours and solid particles lie within this.
- The troposphere is influenced by seasons and jet streams.
Tropopause
- Top most layer of troposphere.
- It acts as a boundary between troposphere and stratosphere.
- This layer is marked by constant temperatures.
Stratosphere
- It lies beyond troposphere, up to an altitude of 50 km from the earth’s surface.
- The temperature in this layer remains constant for some distance but then rises to reach a level of 0°C at 50 km altitude.
- This rise is due to the presence of ozone (harmful ultraviolet radiation is absorbed by ozone).
- This layer is almost free from clouds and associated weather phenomenon, making conditions most ideal for flying aeroplanes. So aeroplanes fly in lower stratosphere, sometimes in upper troposphere where weather is calm.
- Sometimes, cirrus clouds are present at lower levels in this layer.
Ozonosphere
- It lies at an altitude between 30 km and 60 km from the earth’s surface and spans the stratosphere and lower mesosphere.
- Because of the presence of ozone molecules, this layer reflects the harmful ultraviolet radiation.
- The ozonosphere is also called chemosphere because, a lot of chemical activity goes on here.
- The temperature rises at a rate of 5°C per kilometer through the ozonosphere.
Mesosphere
- This is an intermediate layer beyond the ozone layer and continues upto an altitude of 80 km from the earth’s surface.
- The temperature gradually falls to -100°C at 80 km altitude.
- Meteorites burn up in this layer on entering from the space.
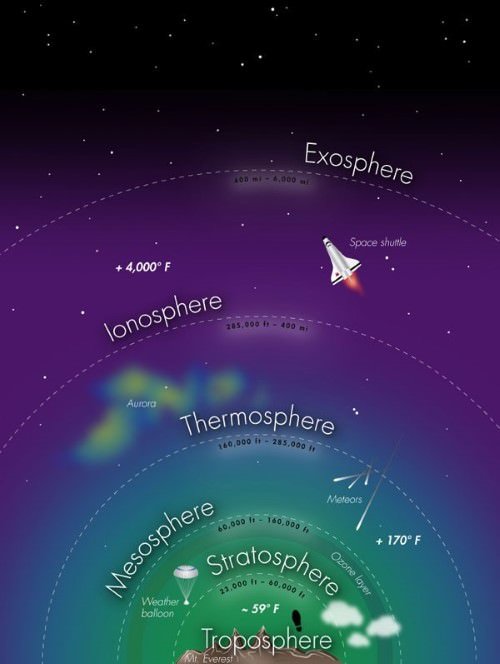
Thermosphere
- In thermosphere temperature rises very rapidly with increasing height.
- Ionosphere is a part of this layer. It extends between 80-400 km.
- This layer helps in radio transmission. In fact, radio waves transmitted from the earth are reflected back to the earth by this layer.
- Person would not feel warm because of the thermosphere’s extremely low pressure.
- The International Space Station and satellites orbit in this layer. (Though temperature is high, the atmosphere is extremely rarified – gas molecules are spaced hundreds of kilometers apart. Hence a person or an object in this layer doesn’t feel the heat)
- Aurora’s are observed in lower parts of this layer.
Ionosphere
- This layer is located between 80 km and 400 km and is an electrically charged layer.
- This layer is characterized by ionization of atoms.
- Because of the electric charge, radio waves transmitted from the earth are reflected back to the earth by this layer.
- Temperature again starts increasing with height because of radiation from the sun.
Exosphere
- This is the uppermost layer of the atmosphere extending beyond the ionosphere above a height of about 400 km.
- The air is extremely rarefied and the temperature gradually increases through the layer.
- Light gases like helium and hydrogen float into the space from here.
- Temperature gradually increases through the layer. (As it is exposed to direct sunlight)
- This layer coincides with space.
Speed of sound follows temperature profile
- This is because speed of sound is directly proportional to temperature as we move away from earth.
Primary References: NCERT Geography, Certificate Physical and Human Geography [Amazon and Flipkart] and Spectrum’s Geography [Amazon and Flipkart].
Last updated on April 25, 2024 1:10 PM













in both thermosphere and ionosphere you have mentioned “This layer helps in radio transmission. In fact, radio waves transmitted from the earth are reflected back to the earth by this layer.”
I think its ionosphere only.
in both thermosphere and ionosphere you have mentioned “This layer helps in radio transmission. In fact, radio waves transmitted from the earth are reflected back to the earth by this layer.”
I think its ionosphere only.
To to easy to learn and to makes goods note
this is really cool so im giving $1,000,000 to everyone who helped team seas
above information is very helpful and relevant.