Polar Vortex & Ozone Hole (Ozone Depletion)
Subscribe to Never Miss an Important Update! Assured Discounts on New Products!
Must Join PMF IAS Telegram Channel & PMF IAS History Telegram Channel
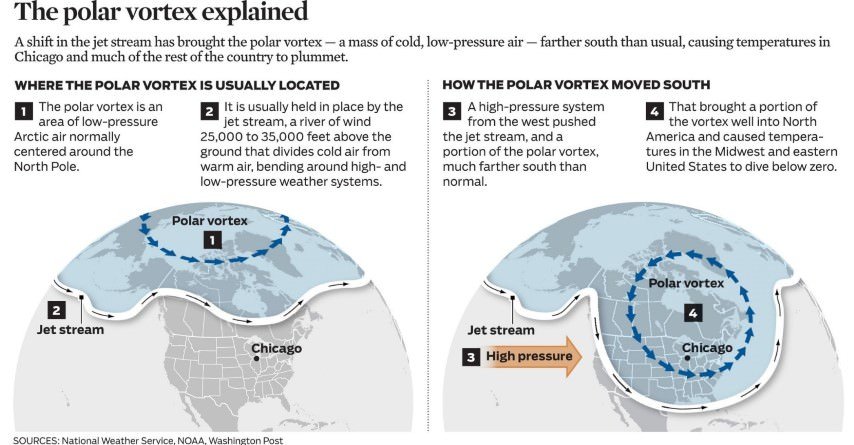
Polar Vortex
- In the previous posts, we have studied about tropical cyclones and extra tropical cyclones (Temperate Cyclone). Here we will study Polar vortex (circumpolar vortex) which is a polar cyclone.
- A polar vortex is a large pocket of very cold air, typically the coldest air in the Northern Hemisphere, which sits over the polar region during the winter season.
- Polar Vortex is a
- Cold;
- Upper tropospheric: sometime extending till the lower levels of stratosphere (At poles, the troposphere extends up to 8-9 km);
- Circumpolar;
- Low pressure;
- Large cyclonic parcel of air [1000 km] (counter-clockwise in the Northern Hemisphere)
- Polar vortex is closely associated with jet streams [Rossby waves].
- It is formed mainly in winter and gets weaker in summer.
- It surrounds polar highs and lie within the polar front (boundary separating the temperate and polar air masses).
Polar Vortex Cold Wave
How Polar Vortex slips towards Midlatitudes,
Breakdown of the polar vortex,
Sudden stratospheric warming,
Polar vortex event.
All the above phrases mean the same – Polar Vortex Cold Wave.
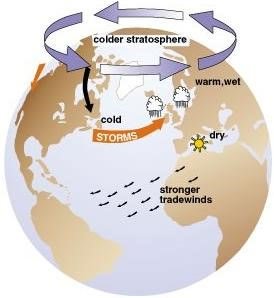
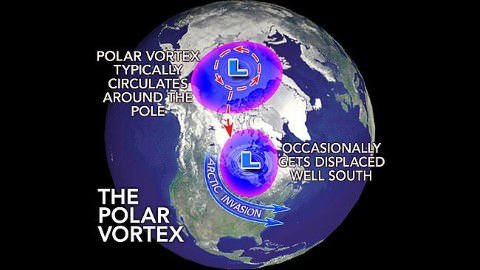
- The polar vortex will remain in its place when the Westerlies along with the polar jet are strong (Strong polar vortex means there is huge temperature contrast between the temperate and polar regions).
- When the polar vortex is weak, it intrudes into the midlatitude regions by buckling the general wind flow pattern. This leads to significant cold outbreaks in the midlatitude regions.
- The vortex is capable of delivering subzero temperatures to the United States and Canada where is occurs the most.
How it slips
- The Polar jet traverses somewhere over 650 N and S latitudes. When the temperature contrast between polar and temperate regions is maximum, the jet is very strong and the meandering is negligible.
- But when the temperature contrast is low (doesn’t mean it’s summer), the jet starts to meander (Rossby waves).
- Meandering jet creates alternating low and high pressure cells. High pressure cells are created below the ridges and the low pressure cells below the troughs (This is because of the upper air circulations created by the jet).
- With severe meandering, the high pressure cells push over to north and displace the polar cyclone from its normal position i.e. the cyclone moves away from the pole and slips into the temperate regions where there is an intense low pressure.
- With the strengthening of the jet, the high pressure cells become weak and retreat to their normal latitudinal positions. With the retreat of the high pressure cells, the polar cyclone moves back to its normal position – poles.
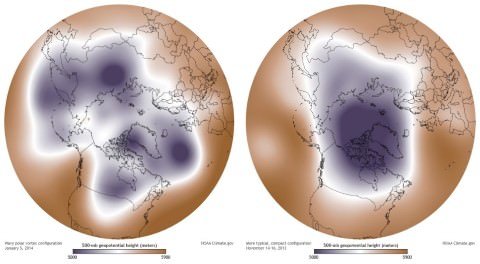
Ozone Hole [Ozone Depletion at South Pole]
- Polar vortex and ozone depletion are two distinct but related phenomena.
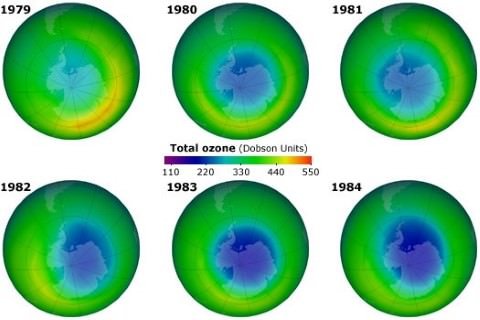
- There is a steady decline of about 4% in the total volume of ozone in Earth’s stratosphere.
- Much larger decrease in stratospheric ozone is observed around Earth’s polar regions.
- Depletion of ozone is due to increase in halocarbons in the atmosphere.
Halocarbon == a compound in which the hydrogen of a hydrocarbon is replaced by halogens like chlorine, bromine, iodine etc.
Halogen == group of reactive non-metallic elements like fluorine, chlorine, bromine, iodine, and astatine.
Halogen atoms like chlorine destroy ozone
- Photodissociation (under the influence of sunlight) of ozone-depleting substances (ODS) like halocarbon refrigerants, solvents, propellants, and foam-blowing agents (CFCs, HCFCs, carbon tetrachloride and trichloroethane, freons, halons) creates free chlorine atoms that destroy ozone.
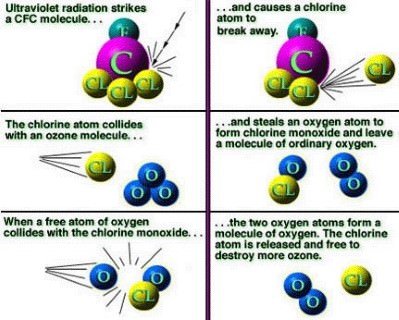

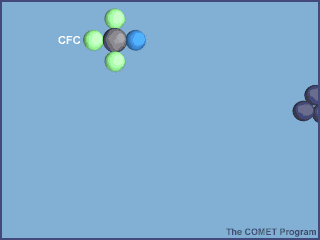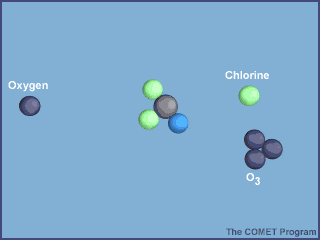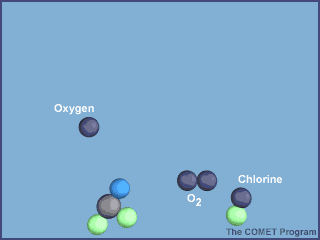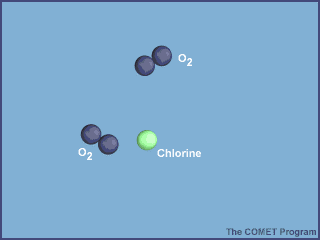
But how does a chlorine atom reach to such high levels of atmosphere?
Polar Stratospheric Clouds (PSCs)
- Extend from 12 km – 22 km above the surface.
- They are nacreous clouds.
Nacreous clouds
|
- Polar Stratospheric Clouds or nacreous clouds contain water, nitric acid and/or sulfuric acid.
- They are formed mainly during the event of polar vertex in winter; more intense at south pole.
- The Cl-catalyzed ozone depletion is dramatically enhanced in the presence of polar stratospheric clouds (PSCs) [Finally this how polar vortex leads to ozone depletion]
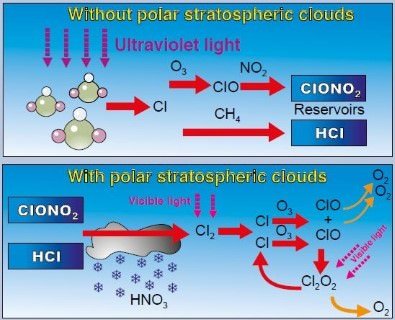
- Polar Stratospheric Clouds convert “reservoir” compounds into reactive free radicals (Cl and ClO).
- These free radicals deplete ozone as shown in the animation below.
- So Polar Stratospheric Clouds accelerate ozone depletion.
Prelims question: The formation of ozone hole in the Antarctic region has been a cause of concern. What could be the reason for ozone depletion at poles?
- Presence of prominent tropospheric turbulence; and inflow of chlorofluorocarbons
- Presence of prominent polar front and stratospheric Clouds and inflow of chlorofluorocarbons
- Absence of polar front and stratospheric clouds; and inflow of methane and chlorofluorocarbons
- Increased temperature at polar region due to global warming
Ozone Depleting Substances
Ozone Depletion is already explained in Geography > Climatology > Polar Vortex
- Ozone depleting substances are man-made gases that destroy ozone once the gases reach the ozone layer.
Ozone depleting substances include:
- chlorofluorocarbons (CFCs)
- hydrochlorofluorocarbons (HCFCs)
- hydrobromoflurocarbons (HBFCs)
- halons (were used in fire extinguishers)
- methyl bromide (was used as fumigant. Fumigation is a method of pest control by suffocating pests with poison)
- carbon tetrachloride (formerly widely used in fire extinguishers, refrigerant and as a cleaning agent)
- methyl chloroform (solvent for organic compounds; used for cleaning metal parts and circuit boards).
They have been used as:
- refrigerants in commercial, home and vehicle air conditioners and refrigerators
- foam blowing agents
- a component in electrical equipment
- industrial solvents
- solvents for cleaning (including dry cleaning)
- aerosol spray propellants
- fumigants.
Chlorofluorocarbons
- Chlorofluorocarbons (CFCs) were widely used as refrigerants until the 1980s.
- They are also considered greenhouse gases that cause climate change.
- There are still CFCs left in older industrial air conditioning and refrigeration systems.
Hydrochlorofluorocarbons
- Hydrochlorofluorocarbons (HCFCs) have been used as a substitute for CFCs.
- They do less damage to the ozone layer than CFCs.
Halons
- Halons are halocarbon gases that were originally developed for use in fire extinguishers.
- Production and consumption of halons ended in 1994 in developed countries
Vienna Convention for the Protection of the Ozone Layer
- The Vienna Convention for the Protection of the Ozone Layer (Vienna Convention) was agreed in 1985.
- It established global monitoring and reporting on ozone depletion.
- It also created a framework for the development of protocols for taking more binding action.
Montreal Protocol on Substances that Deplete the Ozone Layer
- The Montreal Protocol under the Vienna Convention (the protocol) was agreed in 1987.
- It facilitates global cooperation in reversing the rapid decline in atmospheric concentrations of ozone.
- Under the protocol countries agreed to phase out the production and consumption of certain chemicals that deplete ozone.
- Phase out of these substances is required by specific deadlines.
- The Vienna Convention and its Montreal Protocol are the first and only global environmental treaties to achieve universal ratification, with 197 parties.
- As a result of the international agreement, the ozone hole in Antarctica is slowly recovering.
- Climate projections indicate that the ozone layer will return to 1980 levels between 2050 and 2070.
Kigali amendment to Montreal Protocol 2016
- Kigali is the capital city of Rwanda.
- In the 28th meeting of the Parties (2016) to the Montreal Protocol, negotiators from 197 nations have signed an agreement to amend the Montreal Protocol in Kigali.
- The parties are expected to reduce the manufacture and use of Hydrofluorocarbons (HFCs) by roughly 80-85% from their respective baselines, till 2045.
- This phase down is expected to arrest the global average temperature rise up to 0.5 °C by 2100.
- It is a legally binding agreement between the signatory parties with non-compliance measures.
- It came into effect from 1st January 2019.
India’s position
- India has agreed on a lenient schedule as it consumes only 3% of HFCs as compared to the other nations like the USA (37%) and China (25%).
Hydrofluorocarbons (HFCs)
- Hydrofluorocarbons (HFCs) are used since as an alternative to CFCs and HCFCs.
- HFCs do not contribute to ozone depletion.
- However, they are potent greenhouse gases with high global warming potential.
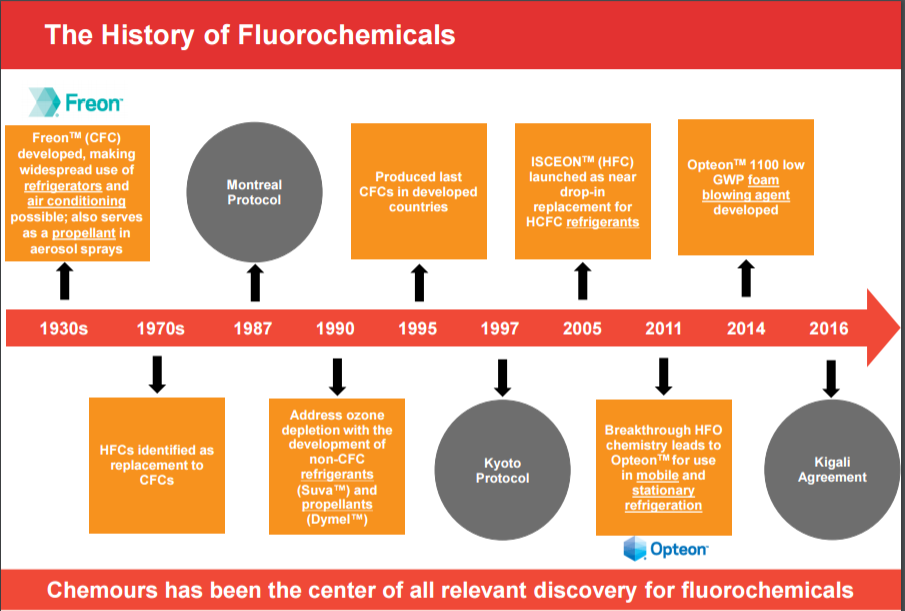
HFO Alternatives to HFCs
- HFO stands for hydrofluoro olefin.
- HFO refrigerants are composed of hydrogen, fluorine and carbon atoms, but contain at least one double bond between the carbon atoms.
Advantages of HFCs
| Fluorochemical | Ozone Depleting Potential | Global Warming Potential |
| CFS | High | High |
| HCFCs | Low | High |
| HFCs | Zero | High |
| HFOs | Zero | Very Low |
The Ozone Depleting Substances Rules
- The rules are framed under the jurisdiction of Environment (Protection) Act.
- These Rules set the deadlines for phasing out of various ODSs, besides regulating production, trade import and export of ODSs and the product containing ODS.
- These Rules prohibit the use of CFCs in manufacturing various products beyond 1st January 2003 except in metered dose inhaler and for other medical purposes.
- Similarly, use of halons is prohibited after 1st January 2001 except for essential use.
- Other ODSs such as carbon tetrachloride and methylchoroform and CFC for metered dose inhalers can be used upto 1st January 2010.
- Further, the use of methyl bromide has been allowed upto 1st January 2015.
- Since HCFCs are used as interim substitute to replace CFC, these are allowed up to 1st January 2040.
Q. Consider the following statements: Chlorofluorocarbons, known as ozone-depleting substances, are used
- in the production of plastic foams
- in the production of tubeless tyres
- in cleaning certain electronic components
- as pressurizing agents in aerosol cans
Which of the statements given above is/are correct?
- 1, 2 and 3 only
- 4 only
- 1, 3 and 4 only
- 1, 2, 3 and 4
Answer: c) CFCs were used as refrigerants, pressurizing agents (foam and aerosol cans) and for cleaning electronic equipment.
Q. Which one of the following is associated with the issue of control and phasing out of the use of ozone-depleting substances?
- Bretton Woods Conference
- Montreal Protocol
- Kyoto Protocol
- Nagoya Protocol
- Bretton Woods Conference established the International Bank for Reconstruction and Development (IBRD) and the International Monetary Fund (IMF).
- Montreal Protocol is an international treaty to protect the ozone layer by phasing out the production of ozone depleting substances. It is legally binding.
- Kyoto Protocol implemented the objective of the UNFCCC to fight global warming by reducing greenhouse gas concentrations in the atmosphere to “a level that would prevent dangerous anthropogenic interference with the climate system”. It is binding on the parties.
- Nagoya Protocol is a supplementary agreement to the 1992 Convention on Biological Diversity (CBD) on “Access to Genetic Resources and the Fair and Equitable Sharing of Benefits Arising from their Utilization”.
Answer: b)